The Interplay Between Thermodynamics and Kinetics in the Solid-state Synthesis of Layered Oxides
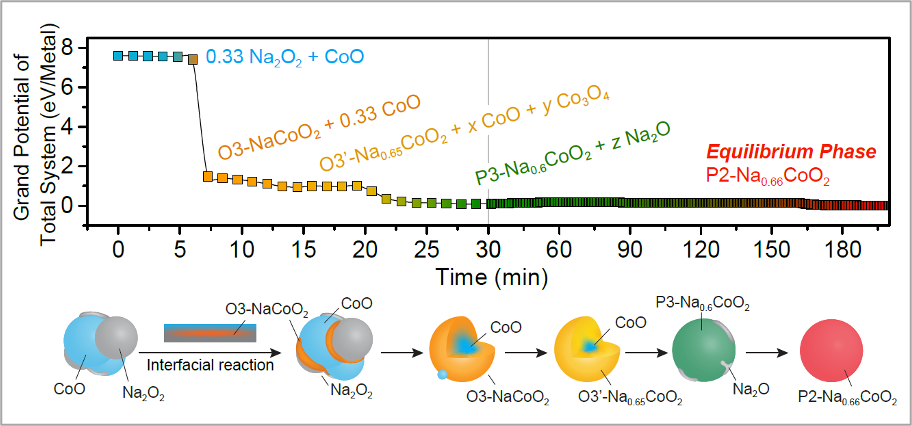
In contrast to the prevailing intuition that solid-state reactions are slow, beamline experiments here show that non-equilibrium phases appear within only a few minutes of starting a ceramic synthesis reaction. These fast-forming intermediates consume most of the thermodynamic reaction energy, resulting in slow transformation kinetics to the stable equilibrium phase. Read full publication here.
Contact Us
Department offices are located in 210 Hearst Memorial Mining Building, in the Northeast corner of campus.
Address:
Department of Materials Science and Engineering
210 Hearst Memorial Mining Building
University of California
Berkeley, CA 94720-1760
Phone: (510) 642-3801
Fax: (510) 643-5792
